सोमाक्लोनल वेरिएशन: फसल सुधार के लिए एक तकनीक
Plant tissue culture procedures give a substitute strategy for vegetative propagation of horticultural crops. Clonal propagation through tissue culture (popularly known as micro propagation) can be acknowledged generally quickly inside a little space. The consistency of individual plants inside a clone population is a significant bit of choice of clonal cultivars in business creation.
However, genetic variations do happen in undifferentiated cells, detached protoplasts, calli, tissues and morphological qualities of in vitro raised plants. In 1981, Larkin and Scowcroft begat an overall term ''somaclonal variation'' for plant variations got from any type of cell or tissue cultures.
Somac1onal variation has been used in an enormous number of plant species such as potato, sugarcane, tobacco, tomato, wheat, rice, brassica and others for different agronomic qualities, for example, disease resistance, plant height, tiller number, maturity and for different physiological and biochemical characteristics.
A few valuable somac1onal variations have been gotten and some of them have been released as cultivars (Hare et al. 2016). Somaclonal variation along these lines seems, by all accounts, to be a significant source of genetic fluctuation. Tissue culture-determined plants show variation which is similar to mutations in tissues and cultured cells.
It has been discussed whether somaclonal variation is the consequence of previous genetic contrasts in physical cells or is initiated by explicit segments of the medium. A few factors, for example, genetic information, explants source, medium synthesis, and period of culture influence somaclonal variation (Bairu et al., 2011).
The main characteristic of somaclonal variation is variations for karyotype, isozyme characteristics and morphology in somaclones may also observe. Calliclone (clones of callus), mericlone (clones of meristem) and protoclone (clones of Protoplast) were produced. Generally heritable mutation and persist in plant population even after plantation into the field.
Mechanism of Somaclonal Variations
1. Genetic (Heritable Variations)
The genetic variation occurs in pre-existing somatic cells of explants and caused by mutations and other DNA changes. These variations occur at high frequency and also change in copy number of DNA sequences is a frequent event in plant tissue cultures.
Amplification of dynamic genes is a typical component for resistance against solid selection yet amplification can likewise happen in cells developing without selection pressure. Decrease or increase in duplicate number of ribosomal genes has been accounted for in numerous species and there are various instances of changes in non-coding DNA.
We have utilized in situ hybridization to recognize variation in telomeric heterochromatin in somacloncs of rye. Changes were seen in ancestors of three regenerants from various cultured undeveloped organisms, one of which included two free amplification occasions. In spite of the fact that point mutations have been recognized in recovered plants, there are barely any cases where these have been dissected at the DNA level.
In one such investigation in maize a solitary regenerant with another electrophoretic type of the protein alcohol dehydrogenase (ADH) was appeared to have a solitary base pair change in exon 6 of the ADH quality.
2. Epigenetic (Non-heritable Variations)
Plants got from in vitro cultures can show phenotypic changes that are not sent through a sexual cycle and are thusly named "epigenetic". In spite of the fact that it is enticing to reject these progressions from genuine somaclonal variation, practically speaking it is difficult to do as such.
In micro propagation and floriculture, for instance, the results of business esteem are the plants that are gotten straightforwardly from the culture and any deviations from the normal phenotype have direct money related outcomes.
Besides, in plants with long age times, for example, palms and trees, there is little chance to set up the heritability of tissue-culture-incited changes. It is in this way of impressive importance to distinguish the system which offers ascend to epigenetic variation in recovered plants (Niwas et al., 2020b).
A change in phenotype without a change in genotype with variations generated during tissue culture, caused by temporary phenotypic changes and occurs at low frequency.
Steps of Somaclonal Variation
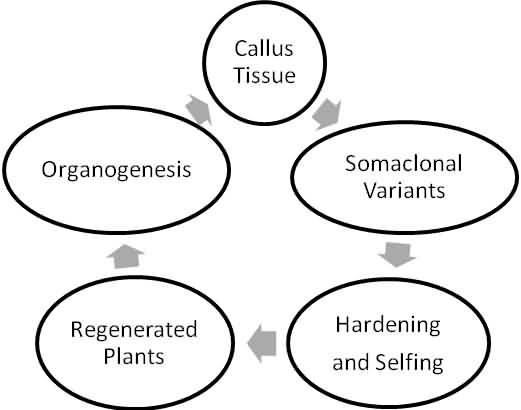
Cause of Somaclonal Variation
There are three main reasons to causes the somaclonal variation.
Physiological causes:
The somaclonal variation developed due to physiological activity such as plant hormone, plant growth regulators, and culture conditions of the media. This type of variation caused when the sample expose a longer time in a particular conditions.
Genetic causes:
The most of the somaclonal variations developed due to genetic reason. The genetic causes govern by changes in chromosomal number (Euploidy, Aneuploidy, polyploidy, monoploidy) chromosomal structure (Deletion, inversion, duplication, translocation), gene mutation (Transition, transversion, insertion, deletion), plasma gene mutation (self-replicating genetic particle postulated to be in the cytoplasm of a cell, as like in mitochondria) and DNA sequences (Changes in DNA, protein) etc.
Biochemical causes:
This is developed by some biochemical elements such as nitrogen metabolism, antibiotics resistance, carbon metabolism and lack of photosynthetic ability.
Table.1: Crop Improvement through somaclonal variation (Niwas et al. 2018 and Niwas et al. 2020a)
| Sl. No. | Plant | Trait improved |
| 1. | Apple (Malusx domestica Borkh.) | Resistance to Erwinia amylovora |
| 2. | Brinjal (Solanum melongena L.) | Stress tolerant somaclone selection |
| 3. | Banana (Musa acuminate L.) | Semi dwarf and resistant to Fusarium wilt |
| 4. | Capsicum (Capsicum annuum L.) | Yellow fruited var. Bell sweet |
| 5. | Potato (Solanum tuberosum L.) | Somaclones for heat tolerance |
| 6. | Rice (Oryza sativa) | Submerge tolerance |
| 7. | Wheat (Triticum aestivum) | Resistance to Bipolaris sorokiniana |
Table.2: Variety developed through somaclonal variation (Ben-Salah and Roath, 1994)
| SN Crop | Variety | Released year | Characteristics | Developed by |
| 1. Sugarcane | Co 94012 (Phule savitri) | 2006 | High yield, red rot and smut diseases resistant | MPKV, Rahuri |
| VSI 434 | 2016 | High cane and sugar yield, high sucrose content and moderately resistant to smut, grassy shoot and red rot diseases | VSI, Pune | |
| 2. Rice | Dama | 1992 | Haploid somaclone, salinity tolerance | Heszky and Simon-Kiss, Hungary |
Advantage of somaclonal variation
The advantages comprise:
(1) It is less expensive than different techniques for genetic manipulation and doesn't require 'containment' strategies.
(2) Tissue culture frameworks are accessible for more plant species than can be controlled by substantial hybridization and transformation right now.
(3) It isn't important to have distinguished the genetic basis of the characteristic, or indeed, in the case of transformation, to have secluded and cloned it.
(4) Novel variations have been accounted for among somaclones, and proof indicate that both the recurrence and dissemination of genetic recombination occasions can be changed by passage however tissue culture.
(5) It helps in crop improvement.
(6) This techniques important for creation of additional genetic variations.
(7) It Increased and improved production of secondary metabolites.
(8) Selection of plants resistant to various toxins, herbicides, high salt concentration and mineral toxicity.
(9) It is also suitable for breeding of tree species.
Somaclonal variation has been best in crops with restricted genetic frameworks (e.g., apomicts, vegetative reproducers) and/or thin genetic bases. In elaborate plants, for instance, the abuse of in vitro-generated variability has become business as usual breeding act of numerous business ventures.
Disadvantage of somaclonal variation
The main disadvantages are:
(1) A serious disadvantage occurs in operations which require clonal uniformity, as in the horticulture and forestry industries where tissue culture is employed for rapid propagation of elite genotypes (Duncan, 1997).
(2) Somaclonal Variations Sometime leads to undesirable results.
(3) Selected variants are random and genetically unstable.
(4) In this works require extensive and extended field trials
(5) It is Not suitable for complex agronomic traits like yield, quality etc.
(6) It may develop variants with pleiotropic effects which are not true.
References
Bairu M W, Aremu A O, Staden J V (2011) Somaclonal variation in plants: causes and detection methods. Plant Growth Regul. 63:147-173.
Ben-Salah H and Roath W W (1994) Somaclonal variation in Cuphea viscosissima Jacq. for plant improvement. Ind Crop Prod. 2:239-244.
Duncan RR (1997) Tissue culture-induced variation and crop improvement. Adv Agron 58:201-240.
Hare K, Mahdi A, Dhurendra S, Udayvir S, Nitesh C, Maliheh E and Radha K S (2016) Somaclonal variations and their applications in horticultural crops improvement. 3. Biotech. 6:54.
Iuliana and Cerasela P (2014) The effect of the ultraviolet radiation on the somaclonal variability for Solanum tuberosum. Rom Biotechnol Lett. 19(3):9339-9344.
Niwas R, Chand G and Azad C S (2018) Evaluation of micronutrients for inhibition of panama wilt disease pathogen (Fusarium oxysporum f. sp. cubense) of banana. Annals of Plant Protection Sciences. 27(1): 81-83.
Niwas R, Chand G and Azad C S (2020a) Evaluation of micronutrients for inhibition of panama wilt disease pathogen (Fusarium oxysporum f. sp. cubense) of banana. Annals of Plant Protection Sciences. 28(1): 47-51.
Niwas R, Chand G and Azad C S (2020b) Morphological and Genetic Diversity of Fusarium spp. associated with Panama wilt disease of Banana in Bihar. Research Journal of Biotechnology. 15(5): 19-23.
Authors:
Ram Niwas1*, Nidhika Rani1, Ankit Kumar Pandey2, Kumar Harsh2, Sampurna Nand Singh2, Gautam Pratap Singh3 and Ankur Kumar Rai2
1Department of Plant Pathology,
2Department of Horticulture (Vegetable & Floriculture),
3Department of Horticulture (Fruit and Fruit Technology),
Bihar Agricultural University, Sabour, Bhagalpur, Bihar 813210, India
Email:
